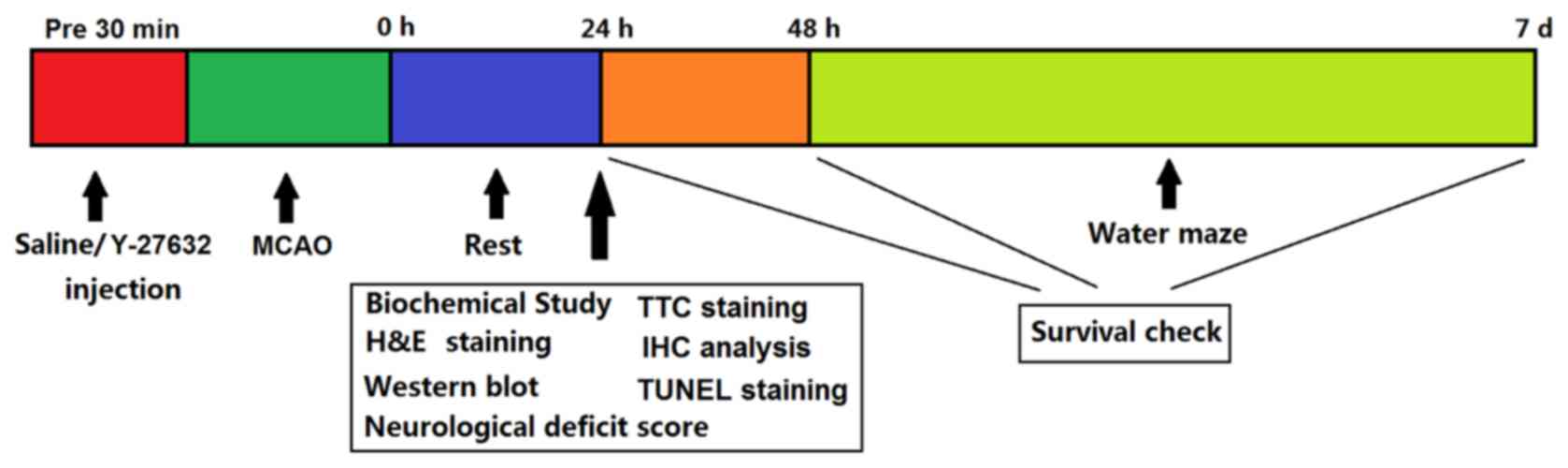
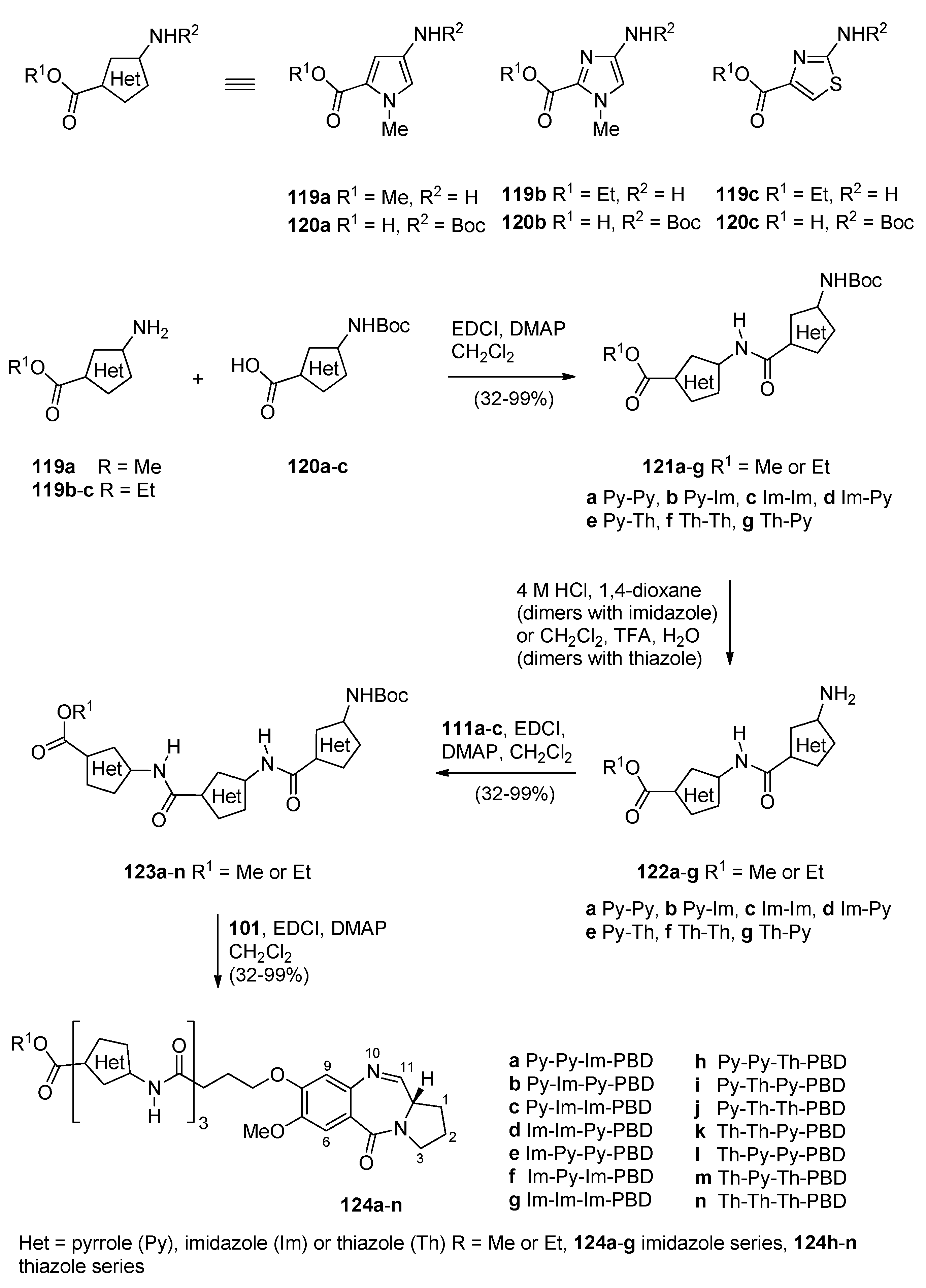
In particular, he should enquire whether the patient has previously experienced adverse reactions to ACTH, Synacthen or other drugs. 4.4 Special warnings and precautions for useīefore using Synacthen, the doctor should make every effort to find out whether the patient is suffering from, or has a history of, allergic disorders (see Section 4.3 “Contra-indications”). asthma) (see Section 4.4 Special warnings and precautions for use), acute psychosis, infectious diseases, peptic ulcer, refractory heart failure, Cushing’s syndrome, treatment of primary adrenocortical insufficiency and adrenocongenital syndrome. Synacthen is contra-indicated in patients with allergic disorders (e.g. Known hypersensitivity to tetracosactide and/or ACTH or to any of the excipients (see section 6.1 List of excipients). For more accurate dosing of other ages, standard body surface area tables should be consulted.Įlderly: There is no evidence to suggest that dosage should be different in the elderly. Thus for children aged 5 to 7 years, approximately half the adult dose will be adequate. Where the 30-minute test has yielded inconclusive results, or where it is desired to determine the functional reserve of the adrenal cortex, a 5-hour test can be performed with Synacthen Depot (see separate Summary of Product Characteristics). Furthermore, a 3-day test with Synacthen Depot may be used to differentiate between primary and secondary adrenocortical insufficiency.Ĭhildren: An intravenous dose of 250micrograms/1.73m 2 body surface area has been suggested. All the plasma samples should be stored in a refrigerator until plasma cortisol level estimation.
if the value 30 minutes after injection is >500 nmol/litre (180 micrograms/litre), adrenocortical function is regarded as normal. The 30-minute Synacthen diagnostic test: This test is based on measurement of the plasma cortisol concentration immediately before and exactly 30 minutes after an intramuscular or intravenous injection of 250micrograms (1ml) Synacthen. Adrenocortical function can be regarded as normal if the post-injection rise in plasma cortisol concentration increases by 200 nmol/litre (70 micrograms/litre), i.e. 4.2 Posology and method of administrationĪdults: This preparation of Synacthen is intended for administration for diagnostic purposes only as a single intramuscular or intravenous dose it is not to be used for repeated therapeutic administration. Therapeutic indicationsĭiagnostic test for the investigation of adrenocortical insufficiency. 3 PHARMACEUTICAL FORMĪ clear colourless aqueous solution for intramuscular injection or intravenous infusion in a 1 mL clear glass ampoule.
Tetracosamide atc4 full#
Tetracosactide acetate 250micrograms per ampoule.įor a full list of excipients, see section 6.1. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Protium 40 mg i.v.SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT.Novartis, Australia Novartis, Canada Novartis, Ghana Novartis, Kenya Novartis, Luxembourg Novartis, Libya Novartis, Nigeria Novartis, Romania Novartis, Sudan Novartis, Tanzania Novartis, Zimbabwe Novartis, Israel Novartis, New Zealand Novartis, Oman Novartis, Portugal Novartis, Russian Federation Novartis, South Africa Swedish Orphan, Sweden

Organon, United Arab Emirates Organon, Bahrain Organon, Cyprus Organon, Egypt Organon, Iraq Organon, Iran Organon, Jordan Organon, Kuwait Organon, Lebanon Organon, Libya Organon, Qatar Organon, Saudi Arabia Organon, Sudan Organon, Syria Organon, Yemenĭaiichi Sankyo, Japan Organon, Hong KongĪlliance, United Kingdom Defiante, Austria Novartis, Australia Novartis, Belgium Novartis, Switzerland Novartis, Denmark Novartis, Ireland Novartis, Italy Novartis, Netherlands Novartis, New Zealand Novartis, Portugal Sigma-tau, Germany Swedish Orphan, Sweden Novartis, Chile Novartis, Luxembourg Novartis, Turkey

